Bats—the only flying mammal—display several additional features that are unique among mammals, such as a long lifespan relative to body size, a low rate of tumorigenesis and an exceptional ability to host viruses without presenting clinical disease. Many emerging bat-borne viruses including SARS-CoV and Ebola virus are highly pathogenic in humans, infected bats show no or minimal signs of disease even when high viral titres are detected in tissues or sera.
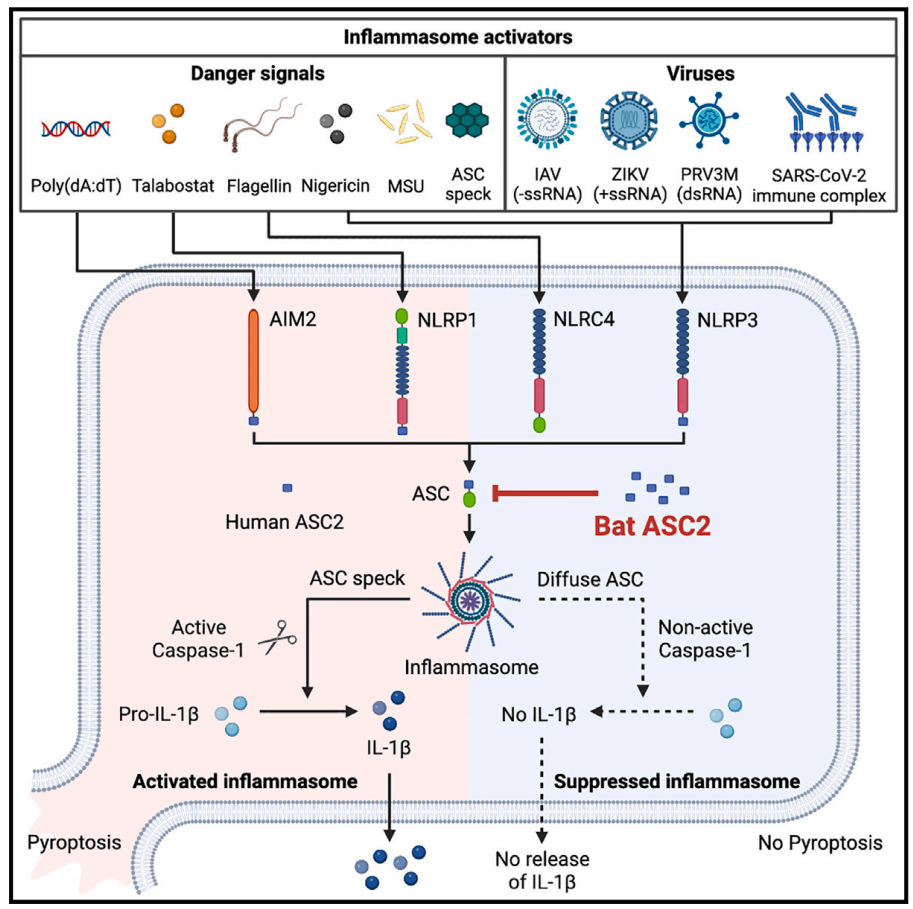
The role of inflammasome signaling in combating inflammatory diseases remains poorly understood. Here, Aaron Irving from ZJE together with Matae Ahn and Prof Wang Lin-Fa's lab from Duke-NUS, Singapore report bat ASC2 as a potent negative regulator of inflammasomes. Bat ASC2 is highly expressed at both the mRNA and protein levels inflammasomes. On the contrary, human ASC2 is poorly expressed or undetectable in most human tissues, with inconsistent functional results of potentiating, inhibiting, or not affecting the inflammasomes. Moreover, while ASC2 and other PYRIN-only proteins were thought to be restricted to primates, this study revealed that ASC2 is present in multiple bat species and highly expressed in bat immune cells. Importantly, in contrast to its human counterpart, bat ASC2 was highly potent in inhibiting both human and mouse inflammasomes. Transgenic expression of bat ASC2 in mice alleviated the severity of sterile peritonitis and reduced the mortality of lethal viral challenge.
It was discovered that the SARS-CoV-2 immune complex triggered NLRP3 inflammasome activation, which was also suppressed by bat ASC2. Additionally, the potential of translation of findings from bat immunity as future therapeutics is also well-demonstrated, it was shown that bat ASC2 suppresses human inflammasomes by interacting with ASC with a higher potency than human ASC2.
This paper demonstrates an important mechanism by which bats limit excessive virus-induced and stress-related inflammation, with implications for their long lifespan and unique viral reservoir status.
Link:https://www.cell.com/cell/fulltext/S0092-8674(23)00333-1







