Chronic lung infections caused by opportunistic pathogens such as Pseudomonas aeruginosa affect the lives and health of people around the world, especially the majority of people who are sick or immunocompromised. Antibiotics are the main treatment option for infections caused by bacteria. However, the treatment of chronic lung infections is more challenging due to the unique biofilm-dominated lifestyle. Compared with planktonic cells, biofilms formed by microbial communities and complex polymer matrices are inherently more resistant to antimicrobial drugs, which is a major factor in clinical relapse. Among the various mechanisms of biofilm resistance, the heterogeneity of microbial biofilms, including chemical gradients and oxygen gradients, has an important impact on drug resistance. For example, cell subpopulations located on the surface of biofilms are metabolically active and grow rapidly. In contrast, cell subpopulations deep in the biofilm are metabolically inactive, grow slowly, or do not grow, corresponding to the spatial heterogeneity of nutrient and oxygen distribution. This means that although antimicrobial agents represented by antibiotics can successfully eliminate bacteria on the surface, they may not be able to fight against metabolically dormant bacteria embedded in biofilms. Therefore, it may be a practical and promising approach to treat difficult biofilm-related chronic infections by combining biofilm metabolic characteristics with biofilm heterogeneity.

To this end, Professor Zhou Min's team from the Second Affiliated Hospital of Zhejiang University School of Medicine/Zhejiang University-University of Edinburgh Institute (ZJU-UoE Institute), Professor Xu Feng from the Department of Infectious Diseases of the Second Affiliated Hospital of Zhejiang University School of Medicine, and Professor Chen Xiaoyuan from the National University of Singapore published an online research paper titled "Wake Biofilm Up to Enhance Suicidal Uptake of Gallium for Chronic Lung Infection Treatment" in the internationally renowned journal "Biomaterials".In this study, we developed a maltohexaose (Glucose polysaccharide, GP)-modified catalase and gallium ion co-loaded delivery polydopamine-coated mesoporous silica (MCPGaGP) nanosystem that can activate the metabolism of dormant bacteria inside the biofilm by converting endogenous hydrogen peroxide in the infection site and biofilm into oxygen, thereby awakening the Pseudomonas aeruginosa (PAO1) biofilm. Activated PAO1 secretes more iron ion acquisition tools, such as siderophores, to enhance the suicidal uptake of gallium ions that mimic iron nutrition. In addition, the internalized gallium ions act as a "Trojan horse" to destroy the biofilm by interfering with the bacterial physiological processes of iron ion acquisition and utilization, biofilm formation, and quorum sensing. Subsequently, the aerosol administration method and the modification of the bacterial-specific transporter maltohexaose enable the delivery of MCPGaGP to the biofilm-induced chronic lung infection area and the targeted release of gallium into the bacteria, providing excellent therapeutic effects. Therefore, we propose an important strategy to awaken biofilms by coordinating biofilm heterogeneity across bacterial nutrition, metabolism, and oxygen gradients and improving the antimicrobial efficacy of gallium-based “Trojan Horse” strategies in biofilm-associated lung diseases.
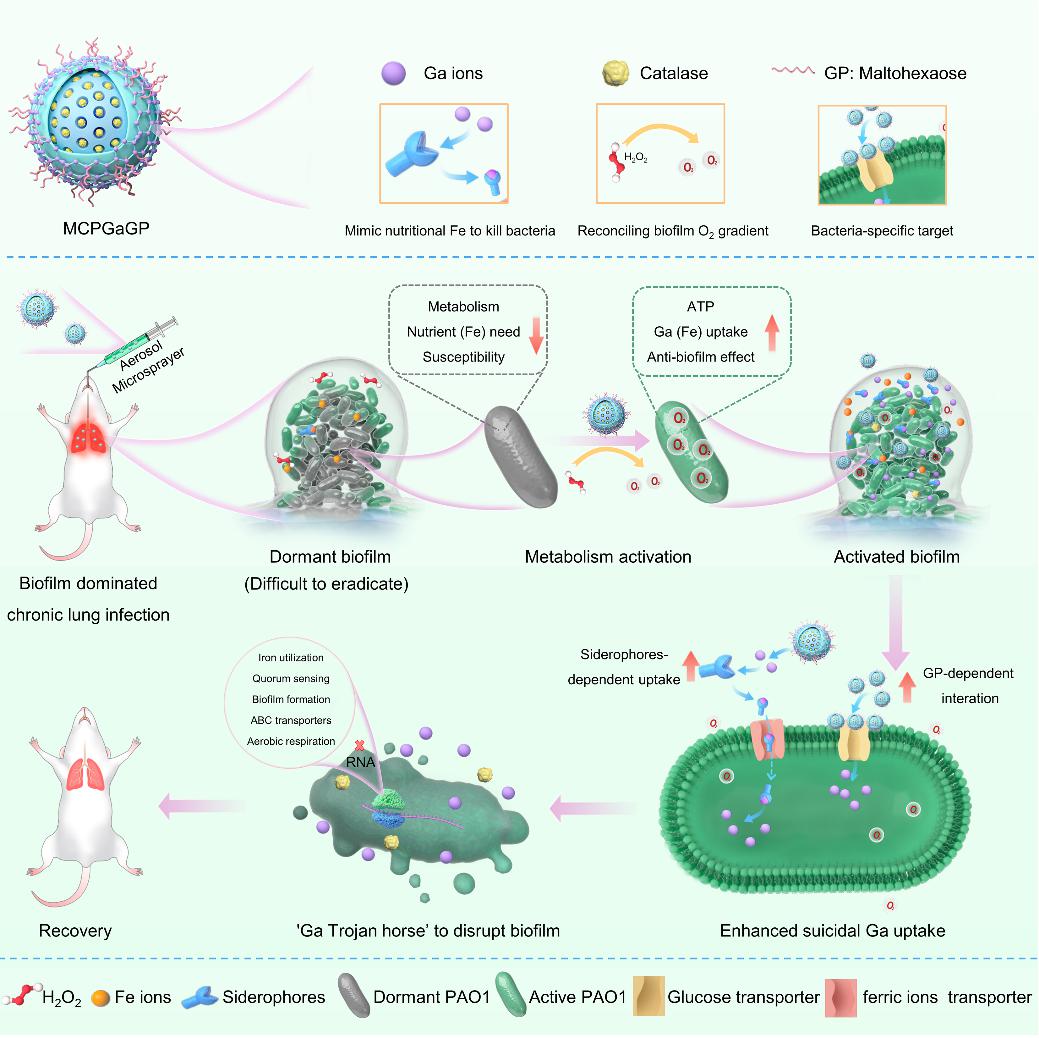
Figure 1: Enhance suicidal uptake of gallium Trojan horses for the treatment of chronic lung infections
Professor Zhou Min of ZJU-UoE Institute and Professor Ouyang Hongwei of Liangzhu Laboratory jointly trained postdoctoral fellow He Jian and Lin Xiuhui, an infectious physician at the Second Affiliated Hospital of Zhejiang University School of Medicine, are the co-first authors of the paper, and Professor Zhou Min of Zhejiang University-Edinburgh Joint College, Professor Xu Feng of the Second Affiliated Hospital of Zhejiang University School of Medicine and Professor Chen Xiaoyuan of Yong Loo Lin School of Medicine/School of Engineering, National University of Singapore are the co-corresponding authors of the paper. The above research was strongly supported by the National Key R&D Program, the National Natural Science Foundation, the Zhejiang Provincial Key R&D Program Project Fund, and the Innovation Institute Medical Transformation Project.
In terms of developing new anti-infection treatment strategies, Professor Zhou Min has conducted in-depth research in recent years on bacterial keratitis, endophthalmitis, and other ophthalmic resistant bacterial infections, refractory fungal keratitis, pneumonia caused by resistant bacteria and difficult-to-heal wounds. In particular, the copper-source ophthalmic nanogel antibacterial method he designed has been clinically tested and has achieved satisfactory clinical antibacterial effects. (Biomaterials 2024; Nano Today 2023; ACS Nano 2023; Adv. Mater. 2022; ACS Nano 2022; Nano Today 2022; ACS Appl. Mater. Interfaces 2022; ACS Nano 2021; Bioact. Mater. 2021; Biomaterials 2021; ACS Nano 2020; Theranostics 2020; Appl. Mater. Interfaces 2019; Appl. Mater. Interfaces 2018;)。







